CE Marking and ARCH clinical trial data position the Liverty™ TIPS Stent Graft to expand treatment options for patients with advanced liver disease
FRANKLIN LAKES, N.J., April 8, 2026 /PRNewswire/ -- BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, today announced application of CE Mark for the Liverty™ TIPS Stent Graft, delivering innovation to portal hypertension care across the European Union. Designed to expand treatment options for patients with cirrhosis and complications of portal hypertension, our next‑generation, adjustable‑diameter device represents the latest advancement by BD in interventional therapies addressing complex liver disease. Safety and effectiveness data from the pivotal ARCH clinical trial evaluating the Liverty™ TIPS Stent Graft will be presented for the first time at the Society of Interventional Radiology (SIR) Annual Scientific Meeting on April 12, 2026.
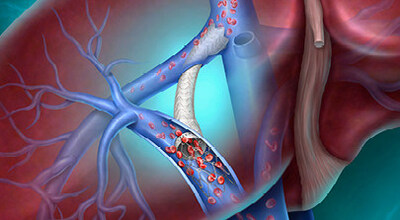
Cirrhosis remains a significant global health challenge, with an estimated 58 million people worldwide living with the disease, according to the latest findings from the Global Burden of Disease study.1 Cirrhosis can lead to portal hypertension, a serious condition that can result in life‑threatening complications such as variceal bleeding and fluid build-up in the abdomen or chest. 2 Transjugular intrahepatic portosystemic shunt (TIPS) procedures help reduce symptoms caused by elevated portal pressure by lowering that pressure and improving blood flow.
The Liverty™ TIPS Stent Graft is engineered to support individualized patient management, offering a 6–10 mm adjustable inner diameter and the broadest range of covered TIPS stent lengths currently available. Key features include a flexible, self-expanding nitinol frame and dual-layer ePTFE encapsulation with carbon impregnation on the inner surface, delivered fully constrained on a triaxial delivery system designed for placement accuracy, control and ease of use.
"For patients living with complications of portal hypertension, access to safe, effective and adaptable treatment options is critical," said Rima Alameddine, worldwide president of Peripheral Intervention at BD. "With CE Marking of the Liverty™ TIPS Stent Graft, we're introducing an innovative, adjustable solution that helps physicians personalize therapy and improve the potential for long‑term outcomes."
BD also announced that data from the ARCH clinical study, a global, prospective, multi‑center, single‑arm clinical study assessing the safety and effectiveness of the Liverty™ TIPS Stent Graft for the treatment of complications from portal hypertension, will be presented for the first time on podium by Global Principal Investigator Prof. Ziv Haskal, MD, at SIR in April.
"The Liverty™ TIPS Stent Graft represents an important advancement in how we care for patients with the serious complications of portal hypertension," said Dr. Haskal. "Its adjustable diameter and delivery design allow physicians to better match the shunt to a patient's unique physiology, a key consideration in safe and effective TIPS creation. The trial provides the first and only prospective data describing the results of small diameter calibrated TIPS for treating all the major complications of portal hypertension. I look forward to presenting the ARCH study results at SIR."
In the United States, the Liverty™ TIPS Stent Graft it is an investigational device limited by U.S. law to investigational use only.
About BD
BD is one of the world's largest pure-play medical technology companies with a Purpose of advancing the world of health™ by driving innovation across medical essentials, connected care, biopharma systems and interventional. The company supports those on the frontlines of healthcare by developing transformative technologies, services and solutions that optimize clinical operations and improve care for patients. Operating across the globe, with more than 60,000 employees, BD delivers billions of products annually that have a positive impact on global healthcare. By working in close collaboration with customers, BD can help enhance outcomes, lower costs, increase clinical efficiency, improve safety and expand access to healthcare. For more information on BD, please visit bd.com or connect with us on LinkedIn at www.linkedin.com/company/bd1/, X @BDandCo or Instagram @becton_dickinson.
________________________________
1 Duo H, You J, Du S, Yu M, Wu S, Yue P, et al. Liver cirrhosis in 2021: Global Burden of Disease study. PLOS One. 2025;20(7):e0328493. doi: 10.1371/journal.pone.032849
2 Carroll A, Boike JR. Tips for TIPS recipients: A patient-focused guidance on TIPS. Clin Liver Dis (Hoboken). 2024;23(1):e0091. doi:10.1097/CLD.0000000000000091
| Contacts: | |
| Media | Investors |
| Matt Marcus VP, Public Relations Matt.Marcus@bd.com | Shawn Bevec SVP, Investor Relations |

Photo - https://mma.prnewswire.com/media/2950341/BD_Delivers_Next_Generation_TIPS_Innovation.jpg
Logo - https://mma.prnewswire.com/media/1479210/BD_Logo.jpg
![]() View original content:https://www.prnewswire.co.uk/news-releases/bd-delivers-nextgeneration-tips-innovation-to-advance-portal-hypertension-care-across-the-european-union-302734832.html
View original content:https://www.prnewswire.co.uk/news-releases/bd-delivers-nextgeneration-tips-innovation-to-advance-portal-hypertension-care-across-the-european-union-302734832.html

Hinweis: ARIVA.DE veröffentlicht in dieser Rubrik Analysen, Kolumnen und Nachrichten aus verschiedenen Quellen. Die ARIVA.DE AG ist nicht verantwortlich für Inhalte, die erkennbar von Dritten in den „News“-Bereich dieser Webseite eingestellt worden sind, und macht sich diese nicht zu Eigen. Diese Inhalte sind insbesondere durch eine entsprechende „von“-Kennzeichnung unterhalb der Artikelüberschrift und/oder durch den Link „Um den vollständigen Artikel zu lesen, klicken Sie bitte hier.“ erkennbar; verantwortlich für diese Inhalte ist allein der genannte Dritte.